Morphology oriented CuS nanostructures: superior K-ion storage using surface enhanced pseudocapacitive effects †
Abstract
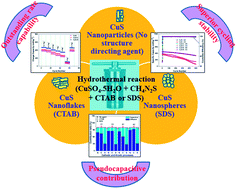
In this work, CuS nanostructures are successfully synthesized by using two different structure directing agents, cetyl trimethyl ammonium bromide (CTAB) and sodium dodecyl sulphate (SDS). For comparison CuS nanostructures are also synthesized without a structure directing agent. The morphologically dependent electrochemical properties such as the capacity, cycling stability, rate capability and capacitive controlled kinetics are comparatively investigated for potassium-ion batteries. The structure directing agents play a vital role in determining the surface area and electrochemical performance of the CuS nanostructures. The CTAB based CuS nanostructure shows a flake like morphology (length 100–150 nm and diameter 5–10 nm) whereas the SDS based CuS exhibits nanospheres with a particle size of 5–15 nm. The capacity, cycling stability and capacitive contribution are higher for the SDS assisted synthesis of the CuS based electrodes as compared to the CTAB assisted CuS and CuS without any agent. At a low current density of 0.1 A g−1, the SDS based CuS electrode exhibits an excellent reversible capacity of 470 mA h g−1. It also exhibits a stable reversible capacity of 451 mA h g−1 with a capacity retention of 73% after 500 cycles at a current density of 0.5 A g−1 (500 mA g−1). The excellent electrochemical performance of the SDS based CuS is attributed to its spherical morphology with a smaller particle size that exhibits a large surface area and the resulting surface dominated pseudocapacitive mechanism enhances the K+ ion storage. The comparative study of these CuS nanostructures can afford significant insights into the design of high performance potassium-ion batteries using hierarchical nanostructures with distinct morphologies.


 Please wait while we load your content...
Please wait while we load your content...