TiO2-mediated visible-light-driven hydrogen evolution by ligand-capped Ru nanoparticles†
Abstract
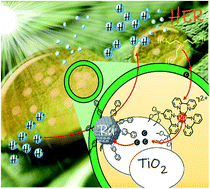
Ru nanomaterials have recently emerged as potential substitutes for classical Pt-based cathodes for the hydrogen evolution reaction (HER). In this regard, nanoparticle surface-functionalization through the so-called organometallic approach is a promising strategy towards synthesizing tailored highly active and durable HER (photo)electrocatalysts with limitless tunability. Herein, efficient (turnover numbers over 480 molH2 molRu−1 and a turnover frequency of 21.5 molH2 h−1 molRu−1; apparent quantum yield of 1.3%) and durable (>100 h) visible-light-driven hydrogen evolution has been achieved at neutral pH with a ternary hybrid nanomaterial combining 4-phenylpyridine-capped Ru nanoparticles (RuPP), TiO2 nanocrystals and [Ru(bpy)2(4,4′-(PO3H2)2(bpy))]Cl2 (RuP) using triethanolamine (TEOA) as a sacrificial electron-donor. Photophysical analysis by means of transient absorption spectroscopy has been performed in order to shed light on the kinetics of the electron transfer events and to identify the rate-determining step of the overall photocatalytic process. TiO2 is shown to have a key role as (1) the support aiding the dispersion of the photocatalyst and limiting its agglomeration under turnover conditions and (2) the electron-transfer mediator enabling efficient electron communication between the catalyst and the anchored molecular photoabsorber. Finally, the evolution and fate of the photocatalyst in long-term HER photocatalysis are thoroughly analyzed.


 Please wait while we load your content...
Please wait while we load your content...