Hydrophilic “bridge” H–C3N4 stabilizing CuO onto graphenes with enhanced energy density for asymmetric supercapacitors
Abstract
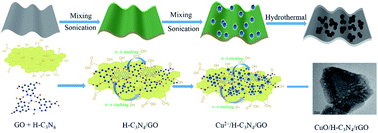
Electroactive species grafted onto graphenes hold great potential as electrode composites for distinguished property supercapacitors. However, the weak bonding and incompatibility between inorganic electroactive species and reduced graphene oxide (rGO) substrates still need to be settled. In this work, H–C3N4 as a novel hydrophilic “bridge” to stabilize CuO anchoring on a graphene substrate was designed to fabricate hydrophilic hybrids. The CuO/H–C3N4/rGO hybrid features a hydrophilic nitrogen-containing functional group, which could exhibit high conductivity and possesses numerous electroactive sites, facilitating fast transfer between electrons and electrolyte ions. In addition, H–C3N4 as a hydrophilic “nano-bridge” could moderate the electronic structural states and firmly combine CuO, the catalytic center, with the substrate rGO, which could enhance the stability of the CuO/H–C3N4/rGO hybrid. Benefiting from these, the as-fabricated CuO/H–C3N4/rGO electrode could exhibit outstanding electrochemical properties with a specific capacitance of 816 F g−1 at a current density of 0.5 A g−1 and 639 F g−1 even at a high current density of 20 A g−1. In addition, the specific capacitance of the CuO/H–C3N4/rGO electrode could maintain 84.38% of the original value after 10 000 cycles. Besides, the asymmetric supercapacitors with the CuO/H–C3N4/rGO hybrid as the anode and activated carbon as the cathode could exhibit a high energy density of 73.85 W h kg−1 at a power density of 0.493 kW kg−1. Moreover, H–C3N4 as “glue” could be used to fabricate certain kinds of active species on different substrates and hold the potential for preparing efficient and robust electrode hybrids for application in various fields.


 Please wait while we load your content...
Please wait while we load your content...