Effects of cation size and concentration of cationic chlorides on the properties of formamidinium lead iodide based perovskite solar cells†
Abstract
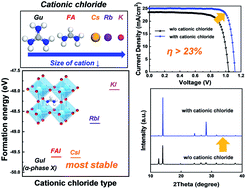
The effect of cationic chlorides has been studied to achieve high quality perovskite films, for high performance perovskite solar cells. However, the role of cationic chloride additives which form mixed-cations on the perovskite structure is still unclear. Investigating the crystal morphology and stability of perovskite films for various sizes or concentrations of cationic chlorides is an essential part of the optoelectronic perovskite study. This paper investigates how cationic chlorides GuCl, FACl, CsCl, RbCl, and KCl, with different cationic sizes and concentrations, affected FAPbI3 perovskite. By forming mixed-cations by the addition of cationic chlorides with appropriate cation size and concentration, based on the tolerance factor, the crystallinity of the perovskite film was improved, achieving a lifetime 2.50 times longer, and a grain size 2.44 times larger. As a result, the efficiency of the perovskite solar cells is increased to 23.28%, and certified as 22.65% at Newport with optimized cationic chlorides. To understand the effect of the cation size and concentration of the cationic chlorides, theoretical analysis of the mechanism by which the cationic chlorides increased the crystallinity was performed using density functional theory calculations. The results revealed the effect of the various cationic chlorides on perovskite phase structures and matched the experimental results.


 Please wait while we load your content...
Please wait while we load your content...