Synthesis and electrochemical performance of Co1−xNixS QDs as electrode materials for high performance supercapacitors†
Abstract
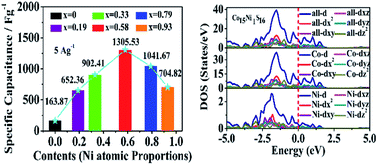
Co1−xNixS quantum dots (QDs), which exhibit outstanding electrochemical properties, are successfully synthesized by a solution diffusion synergistic capture doping method. A larger surface area of Co1−xNixS QDs, which can provide more electroactive sites and promote ion exchange in the electrolyte solution, is beneficial to improve their electrochemical performance. Co1−xNixS (x = 0.58) QDs as a high-performance supercapacitor electrode for supercapacitors deliver not only a large specific capacitance of 1305.53 F g−1 even at 5 A g−1, but also fine rate properties with 729.81 F g−1 at 50 A g−1, as well as excellent cycling stability with a capacitance retention of nearly 94.89% after 10 000 circulations in a 6 M KOH electrolyte. The electrochemical performance of Co1−xNixS electrodes is superior to that of CoS electrodes. The specific capacitance of Co1−xNixS QDs presents a parabolic trend with the increase of Ni doping concentration. The excellent electrochemical properties of Co1−xNixS QDs can be attributed to Ni atom doping, which can significantly enhance electron state density in the 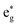 band of both Co and S. On the one hand, the impurity levels introduced by Ni doping can effectively improve the ability of charge storage. On the other hand, Ni atom doping increases the density of Co's and S's empty electron states and leads to the increase of the specific capacitance of Co1−xNixS QDs. A theoretical calculation is carried out to systematically explain the origin of the specific capacitance of Co1−xNixS QDs. The Co1−xNixS electrode with outstanding electrochemical performance has the potential to meet the requirements of practical electrochemical energy storage applications.
band of both Co and S. On the one hand, the impurity levels introduced by Ni doping can effectively improve the ability of charge storage. On the other hand, Ni atom doping increases the density of Co's and S's empty electron states and leads to the increase of the specific capacitance of Co1−xNixS QDs. A theoretical calculation is carried out to systematically explain the origin of the specific capacitance of Co1−xNixS QDs. The Co1−xNixS electrode with outstanding electrochemical performance has the potential to meet the requirements of practical electrochemical energy storage applications.


 Please wait while we load your content...
Please wait while we load your content...