Catalytic mechanism for selective hydrogen production based on formate decomposition with polyvinylpyrrolidone-dispersed platinum nanoparticles†
Abstract
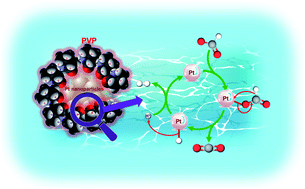
Formic acid (HCOOH) has attracted considerable attention as a hydrogen carrier because it has low toxicity and is easy to handle in aqueous solutions. In this article, the mechanism of H2 production due to HCOOH decomposition with Pt nanoparticles dispersed by polyvinylpyrrolidone at room temperature was investigated. The mechanism was discussed based on the identification of the reactive species by the pH dependence of H2 production from HCOOH and the bonding form of formate on Pt using IR spectroscopy. From these results, we suggested that the possible catalytic mechanism of HCOOH decomposition into H2 and CO2 occurred via the β-hydrogen elimination of CO2, the rate-limiting step, with Pt-PVP. Moreover, the temperature dependence of H2 production from HCOOH was investigated in order to estimate the activation energy. The activation energy of the HCOOH decomposition into H2 and CO2 with Pt-PVP as the catalyst was calculated to be 41 kJ mol−1, which indicated a reduction of about 10% compared to that for the system without any catalyst. In addition, the causes for the deactivation of the catalyst were investigated via transmission electron microscopy and activity evaluation in various conditions. Pt-PVP was deactivated, whereas it was very durable under high-temperature conditions with no CO emission in the gas phase. We propose that the clarification of the H2 production mechanism can provide guidelines to develop new catalysts with high activity for HCOOH decomposition.


 Please wait while we load your content...
Please wait while we load your content...