Three-dimensional flower-like nickel phyllosilicates for CO2 methanation: enhanced catalytic activity and high stability†
Abstract
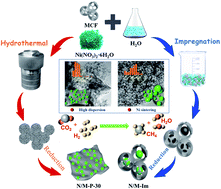
To simultaneously enhance both the activity and stability of the supported Ni catalysts for a high-temperature reaction is a great challenge in catalysis. Addressing this problem, three-dimensional flower-like nickel phyllosilicates were facilely prepared by a hydrothermal method through the reaction between mesostructured cellular foam silica (MCF) and nickel nitrate for CO2 methanation. These Ni-containing phyllosilicates samples are characterized by various techniques, such as nitrogen adsorption, X-ray diffraction, scanning electron microscopy, transmission electron microscopy, H2 temperature programmed reduction, H2 temperature programmed desorption, CO2 temperature programmed desorption and X-ray photoelectron spectroscopy. Our results showed that the nickel phyllosilicate became more pronounced and denser with the increase of hydrothermal time, leading to the enhanced interaction between metal and silicate with the Ni contents in the high level of 20.05–32.05 wt%. In addition, the particle size of Ni is small (<5 nm) after the reduction at 700 °C, ascribed to the formation of nickel phyllosilicate. The phyllosilicate catalyst with the hydrothermal time of 30 h (N/M-P-30) shows the highest catalytic activity, which achieves the maximum CO2 conversion of 79.5% at 450 °C, 0.1 MPa and a high weight hourly space velocity (WHSV) of 60 000 mL g−1 h−1. In the 100 h-lifetime test under the condition of 450 °C, 0.1 MPa, 60 000 mL g−1 h−1 and 6 h-hydrothermal treatments at 600 °C in 100% steam, N/M-P-30 shows high stability without Ni sintering and collapse of pores as well as the decrease of catalytic activity after two stability tests, which is attributed to the special properties of three-dimensional flower-like nickel phyllosilicates and the enhanced interaction between Ni species and silicate.


 Please wait while we load your content...
Please wait while we load your content...