A novel strategy to simultaneously tailor morphology and electronic structure of CuCo hybrid oxides for enhanced electrocatalytic performance in overall water splitting†
Abstract
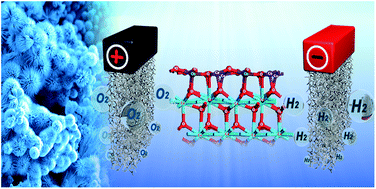
To improve the electrocatalytic performance of CuCo hybrid oxides, here we report a novel and facile strategy to simultaneously tailor the morphology and electronic structure of CuCoOx by incorporation of CeO2. The as-synthesized CeO2-CuCoO/NF shows urchin-like morphology that is composed of numerous nanofibers, endowing the surface with superaerophobicity and massive active sites. Density functional theory calculation elucidates the crucial role of CeO2 in boosting the electron transfer and decreasing the free energy of intermediates during water splitting. As expected, CeO2-CuCoO/NF shows attractive bifunctional electrocatalytic activity with overpotentials of 266 and 93 mV at 10 mA cm−2 for oxygen and hydrogen evolution reactions in 1 M KOH, respectively, which are superior to most reported catalysts. A water-splitting electrolyzer consisted of CeO2-CuCoO/NF electrodes as both anode and cathode displays better performance than the noble-metal counterpart IrO2/NF‖Pt/C/NF. The present work undoubtedly sheds new insight on designing advanced bifunctional catalysts for practical water splitting.


 Please wait while we load your content...
Please wait while we load your content...