Sulfur vacancies promoting Fe-doped Ni3S2 nanopyramid arrays as efficient bifunctional electrocatalysts for overall water splitting†
Abstract
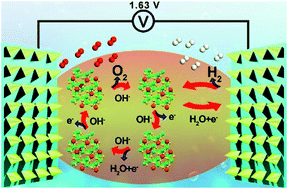
The development of cost-effective bifunctional catalysts for the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) is critical to realize the commercialization of electrocatalytic water splitting. Herein, an effective defect engineering strategy, Ar plasma treatment, was adopted to promote both the HER and OER activity of pre-synthesized Fe-doped Ni3S2 nano-pyramid arrays on nickel foam. The abundant sulfur vacancies endow the Fe-doped Ni3S2 nano-pyramid arrays with a low overpotential of 171 mV at 10 mA cm−2 and a small Tafel slope of 49 mV dec−1 to drive the OER in 1.0 M KOH. Density functional theory (DFT) calculations reveal that sulfur vacancies together with Fe doping can effectively regulate the electronic structure of Ni3S2, resulting in increased electrical conductivity and a reduced energy barrier, as well as enhanced reaction kinetics through weakening the binding strength between active sites and intermediates. Such defect-rich hetero-metal doped catalyst also delivers enhanced HER activity. As a result, a cell assembled using defect-rich Fe-Ni3S2 nano-pyramid arrays as both the anode and cathode needs a low applied voltage of 1.63 V to reach a current density of 10 mA cm−2, and exhibits outstanding long-term stability up to 60 h.


 Please wait while we load your content...
Please wait while we load your content...