Enhanced alkaline hydrogen evolution performance of ruthenium by synergetic doping of cobalt and phosphorus†
Abstract
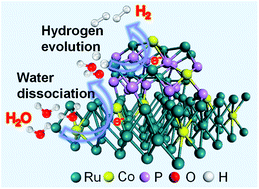
Highly active and stable electrocatalysts that can work well in alkaline media are essential for industrial water electrolysis. Herein, we have reported the growth of P and Co co-doped Ru hybrids (RuCo–P) on carbon cloth (CC) through a hydrothermal treatment and the following phosphorization. The modification of Ru by Co and P doping can tune the electronic structure for efficient water dissociation and hydrogen desorption in alkaline solution. The resulting RuCo–P/CC achieves a record low overpotential of ∼4 mV to reach a current density of 10 mA cm−2 for the hydrogen evolution reaction (HER) in 1 M KOH solution, and also shows a small Tafel slope (43 mV dec−1) and outstanding long-term stability. The activity of RuCo–P/CC is significantly superior to that of the commercial Pt/C benchmark and other carbon cloth-supported transition metal-based catalysts. The remarkable alkaline HER performance of RuCo–P/CC could be attributed to the synergistic doping of both Co and P that can effectively reduce the water dissociation barrier and optimize the hydrogen adsorption free energy, respectively.


 Please wait while we load your content...
Please wait while we load your content...