A novel rechargeable bromine-ion battery and the induction of bromine ions on metal electrodes
Abstract
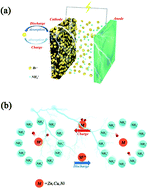
In recent years, bromine has been widely used in batteries due to its higher theoretical specific capacity, but unfortunately, no battery has been reported based on the absorption/desorption principle of a single bromine ion. In our work, the toxicity of bromine in battery applications has been completely solved. A rechargeable bromine-ion battery (BIB) based on the principle of bromine ion absorption/desorption has been proposed for the first time, and the induction effect of bromine ions on metal electrodes has also been described. Cheap carbon black (cb) is used as the positive electrode material, and zinc, copper or nickel is used as the BIB negative electrode. When zinc foil is used as the negative electrode of the BIB, the specific capacity reaches 175 mA h gcb−1 under a charge and discharge current density of 2 A g−1, and the specific capacity is still 141 mA h gcb−1 after 3000 cycles. When copper foil or nickel foil is used as the negative electrode, the capacity is 85 mA h gcb−1 after 500 cycles. Using X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR) and X-ray photoelectron spectroscopy (XPS) characterization tests, it is verified that the bromine ions exhibit absorption/desorption in the carbon material instead of undergoing a redox reaction during the charging and discharging process.


 Please wait while we load your content...
Please wait while we load your content...