Adjusting the active sites of Cu and ZnO by coordination effect of H3BTC and its influence on enhanced RWGS reaction†
Abstract
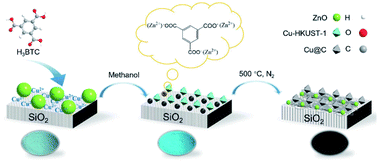
Converting CO2 to CO via the reverse water gas shift (RWGS) reaction is an important route to reducing greenhouse gas emissions, and catalysts using copper and zinc as active components have been extensively studied for this reaction. Herein, the coordination and acid etching of trimesic acid (H3BTC) were used to assemble and regulate the Cu and ZnO components in the catalyst. First, Cu-HKUST-1 was synthesized by the self-assembly of H3BTC and copper ions, which limited the sintering and agglomeration of Cu nanoparticles in the subsequent carbonization and self-reduction process. Second, ZnO particles were etched by H3BTC to form the complex of H3BTC with Zn and to redisperse Zn species. Under the dual action of H3BTC, carbon-coated bimetal nanoparticles with a size of 2–6 nm were obtained on the fumed silica after pyrolysis at high temperature. The prepared Cu@C–ZnO/SiO2 catalyst can achieve 31.4% CO2 conversion and 99% CO selectivity in RWGS reaction in a fixed bed reactor at 400 °C and 3 MPa. Besides, we also found that the molar ratio of Cu/Zn, metal components, and the introduction order of H3BTC all affected the structure and composition of the catalyst, thus affecting the catalytic activity. This study provides an excellent strategy for the design and synthesis of catalysts containing copper and zinc components.


 Please wait while we load your content...
Please wait while we load your content...