Hexanedioic acid mediated in situ functionalization of interconnected graphitic 3D carbon nanofibers as Pt support for trifunctional electrocatalysts†
Abstract
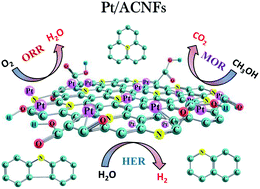
A unique approach of in situ functionalization has resulted in the uniform dispersion of Pt nanoparticles on the surface of hexanedioic acid modified electrospun 3D carbon nanofibers (ACNFs). Unlike the traditional mineral acid based oxidation protocols, a facile single-step methodology without any post-treatments is adopted for the carboxylic functionalization of the carbon support. The interconnected 3D nanostructure with an optimal introduction of metal-binding surface functional groups promotes the uniform dispersion of Pt nanoparticles. The resultant Pt-loaded Pt/ACNFs are used for various electrocatalytic reactions. The Pt/ACNFs exhibit excellent electrocatalytic activity with multifunctional ability, promoting the oxygen reduction reaction (ORR) via an efficient 4e− transfer, providing a large electrochemical surface area (119.21 m2 g−1) and superior mass activity (684.57 A g−1) for the methanol oxidation reaction (MOR), and demanding minimal overpotential (50 mV) and Tafel slope (35 mV dec−1) for the hydrogen evolution reaction (HER). Thus, the superior electrocatalytic performance of the prepared Pt/ACNFs is definitely attributable to the uniform binding of Pt metal particles with the carefully grafted –COOH functional groups.


 Please wait while we load your content...
Please wait while we load your content...