Investigating high-performance sulfur–metal nanocomposites for lithium batteries†
Abstract
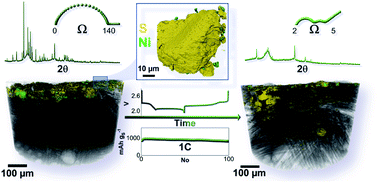
Herein, for the first time, we study the reversible conversion in a lithium cell of a novel sulfur–metal nanocomposite by combining X-ray computed tomography data at the micro- and nanoscales with the electrochemistry. The electrode is obtained at mild temperatures according to an alternative approach, including metal nanoparticles of either tin or nickel in bulk molten sulfur in the corresponding weight ratio of 85 : 15. We show that this pathway leads to the formation of high-performance electrodes, matching the state-of-the-art results obtained from the best carbonaceous composites. Indeed, lithium–sulfur (Li–S) cells at a working voltage of about 2.2 V ensure sulfur-mass-referred capacity approaching 1400 mA h g−1 at a C/3 rate and 740 mA h g−1 at a rate as high as 3C (1C = 1675 mA h g−1), with a coulombic efficiency close to 100% and stable cycling trends over 100 cycles. High-resolution imaging sheds light on the characteristic morphological features of the electrode allowing these remarkable performances, and reveals the beneficial effects of the incorporation of metal nanoparticles within the sulfur phase. The various investigation techniques, with a particular focus on three-dimensional imaging, suggest sulfur electrodeposition upon charging, preferentially adjacent to the electron-conductive centers within the electrode support as well as that on metal clusters. A massive microstructural reorganization is observed during the first cycle in lithium cells with concomitant remarkable enhancements in the electrode charge transfer and variation in the reaction potentials. This process is accompanied by substantial electrode amorphization and migration of the active material toward the current-collector bulk. The results obtained in this work, as well as a comprehensive study with an ad hoc design for sulfur electrodes, suggest alternative strategies for ultimately achieving actual Li–S cell improvement.


 Please wait while we load your content...
Please wait while we load your content...