Are MXenes suitable as cathode materials for rechargeable Mg batteries?†
Abstract
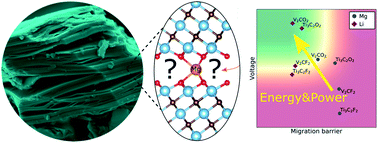
MXenes, a type of two-dimensional transition metal carbide/nitride, have received significant attention for electrochemical storage applications. With substantial capacities predicted for Mg ions, MXenes have been considered a promising candidate for cathode materials for rechargeable Mg batteries (RMBs). While there have been reports demonstrating reversible capacities of 100–200 mA h g−1 for MXene-based cathodes, the understanding of the reactions occurring is limited. Here, we investigate unsolvated Mg2+ intercalation into multi-layered, non-delaminated MXene particles by electrochemical characterization of two common MXenes (Ti3C2Tx and V2CTx) with various electrolytes and at elevated temperatures, complemented by DFT calculations. Our experimental results show poor reversible capacities (<5 mA h g−1) both at room temperature and at 60 °C, indicating that no intercalation of Mg ions occurs during cycling. DFT simulations show that Ti3C2T2 with fluorine and hydroxyl termination groups has a negative average voltage for Mg2+ and reveal high migration barriers for oxygen termination groups, supporting the experimental results. The DFT calculations also reveal that upon magnesiation in multi-layered V2CF2 and V2C(OH)2, MgF2 or MgH2 is formed rather than the electrochemical intercalation of Mg2+. However, multi-layered V2CO2 is found to have both a relatively high theoretical average voltage (∼1.5 V) and a comparatively low migration barrier of 480 meV, allowing for Mg2+ intercalation and RMBs with energy densities comparable to Li-ion batteries. The findings emphasize the need to fully control the MXene termination groups. The possible reasons for the unsatisfactory and deviant electrochemical performance reported in this work and in the literature are critically reviewed to assess the feasibility of MXene cathodes for energy-dense RMBs.


 Please wait while we load your content...
Please wait while we load your content...