Effects of lithium dendrites on thermal runaway and gassing of LiFePO4 batteries
Abstract
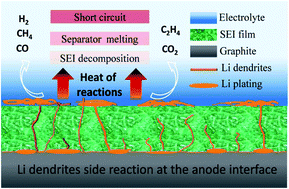
Safety is the key requirement for large-scale applications of lithium-ion batteries, but lithium dendrites challenge the safe operation of lithium-ion batteries with graphite anodes. In this paper, the electrochemical properties of pouch LiFePO4/C batteries with a 10 year calendar life were studied at different temperatures and different current densities at −10 °C. After cycling under different conditions, the thermal safety of the batteries was studied using an Accelerating Rate Calorimeter (ARC) and aging at elevated temperatures was performed to investigate the thermal abuse behavior of the batteries. The results show that the main reason for the decline of electrochemical performance is the increase of internal resistance and the formation of non-electrochemically active lithium dendrites on the anode surface during the low-temperature cycling. Exothermic reaction and gassing are the main ways by which lithium dendrites affect battery safety. Lithium dendrites reduce the temperature of self-heating (Tonset) and the temperature of thermal runaway (TTR), which is from 170 °C to about 100 °C. Electrolytes react with lithium dendrites violently at elevated temperatures to produce gases, which makes the internal pressure of the batteries increase continuously, causing safety problems such as battery explosion and electrolyte leakage. The growth of lithium dendrites destroys the thermal stability of SEI films. Moreover, the thermal side reaction between lithium dendrites and electrolytes accelerates the decomposition of SEI films, reducing the thermal runaway temperature (TTR) of the battery.


 Please wait while we load your content...
Please wait while we load your content...