Tiny amounts of fluorinated carbon nanotubes remove sodium dendrites for high-performance sodium–oxygen batteries†
Abstract
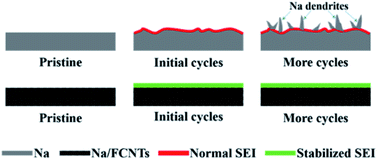
Metallic sodium batteries are regarded as a promising alternative to metallic lithium batteries due to the abundance of Na resources. However, similar to Li, metallic Na suffers from severe dendrite growth during cycling, which hinders its practical applications. Here, we report a Na composite anode composed of metallic Na and tiny amounts of fluorinated carbon nanotubes (FCNTs, 1–1.5 wt%). Compared with bare Na, the Na/FCNT electrode exhibits lower polarization voltage, lower surface resistance, and longer cycle life. The Na/FCNT electrode can sustain stable cycling at 2 mA cm−2 with a capacity as high as 5 mA h cm−2 for 1700 h. Scanning electron microscope (SEM) and in situ optical microscope observations confirm dendrite-free stripping/plating of metallic Na during cycling. The inhibition effect of Na dendrites can be attributed to the NaF-rich solid electrolyte interface (SEI) layer on the Na surface which is in situ formed from the reactions between Na and FCNTs. A Na–O2 cell with the Na/FCNT anode can be stably cycled for 112 cycles at 400 mA g−1 with a limited capacity of 1000 mA h g−1, while that with the bare Na anode can be stably cycled only for 62 cycles. The obviously enhanced cycling stability of the cell with the Na/FCNT anode is due to the protective effect of the in situ fabricated NaF-rich SEI layer.


 Please wait while we load your content...
Please wait while we load your content...