The fascinating supercapacitive performance of activated carbon electrodes with enhanced energy density in multifarious electrolytes†
Abstract
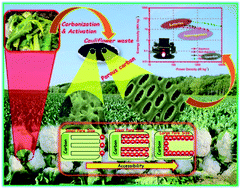
The market for supercapacitors is rapidly growing due to their high power capability, but their energy is low compared with batteries. However, their poor energy density can be modulated with redox additive electrode materials either by decoration or composite fabrication. In addition, the energy density of the supercapacitors could be increased using the redox reaction in the electrolyte itself. Herein, a high-energy supercapacitor is demonstrated using activated high surface area carbon derived from cauliflower and a redox additive electrolyte. The activated carbon shows mesopores with a specific surface area of ∼1500 m2 g−1, and a maximum specific capacitance of 310 F g−1 is achieved in a two-electrode symmetric configuration using 1 M H2SO4 electrolyte. However, the fabricated device shows high specific capacitances of 78 and 92 F g−1 in non-aqueous organic and ionic liquid electrolytes with energy densities of 17 and 29 W h kg−1, respectively. The energy density of the supercapacitor device is further enhanced using the novel redox additive 0.01 M KI in Na2SO4 electrolyte. It shows a specific capacitance of 520 F g−1 and a maximum energy density of 58.5 W h kg−1, which is very high and comparable to that of lithium-ion batteries.


 Please wait while we load your content...
Please wait while we load your content...