Surfactant decorated hydrotalcite-supported polyoxometalates for aerobic oxidation of 5-hydroxymethylfurfural and monosaccharides†
Abstract
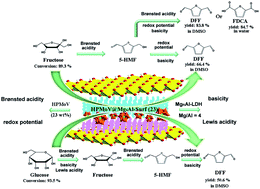
Trifunctional catalysts based on polyoxometalate (POM) and surfactant modified MgAl-layered double hydroxide (LDH) were synthesized and evaluated for aerobic oxidation of 5-hydroxymethylfurfural (5-HMF) and monosaccharides. H5PMo10V2O40@MgnAl-Surf (wt%) (abbreviated as HPMoV@MgnAl-Surf (wt%), n represents the molar ratio of Mg to Al) presented tunable redox potential, Brønsted acidity, Lewis acidity, and basicity upon changing the molar ratio of HPMoV to MgnAl-Surf and also n values. HPMoV@Mg4Al-Surf (23) was found to be the most active and exhibits 88.6% selectivity to 2,5-diformylfuran (DFF) with 94.6% conversion of 5-HMF in dimethyl sulfoxide (DMSO), while presented 90.9% selectivity to 2,5-furandicarboxylic acid (FDCA) with 93.2% conversion in water. The existence of a surfactant being covalently bonded on Mg4Al-LDH provided a hydrophobic surrounding for concentrating the reactants and repelling the product, and promoted the mass transfer within the inter-layer gallery. Meanwhile, higher yields of DFF of 66.4% and 50.6% were achieved directly from fructose and glucose, and were attributed to the suitable balancing of acidity and basicity in multifunctional catalysts. The catalytic mechanism for glucose oxidation on HPMoV@MgnAl-Surf was studied in detail to determine the triple-functional sites on the pathway. Also, the oxidation of 5-HMF, fructose and glucose was achieved under atmospheric pressure of O2, showing the wide availability of HPMoV@Mg4Al-Surf (23). HPMoV@Mg4Al-Surf (23) showed good stability and durability for being reused ten times without any leaching of HPMoV from Mg4Al-Surf due to HPMoV being embedded by the surfactant.


 Please wait while we load your content...
Please wait while we load your content...