The synergetic effect of N, S-codoped carbon and CoOx nanodots derived from ZIF-67 as a highly efficient cocatalyst over CdS nanorods†
Abstract
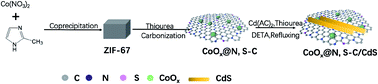
Earth-abundant transition metal cocatalysts are promising rare and noble metal alternatives for efficient photocatalytic hydrogen evolution. Herein, a novel composite of ZIF-67 derived CoOx nanodots confined in a N, S co-doped graphitic nanosheet co-catalyst (CoOx@N, S-C) was successfully constructed via a one pot carbonization process. Through loading onto photosensitizer of CdS nanorods, the obtained CoOx@N, S-C/CdS composite exhibited an unprecedented H2 production rate of 40.1 mmol g−1 h−1, which is 57, 20 and 1.7 times higher than that of CdS, N, S-C/CdS and CoOx/CdS, respectively. It was demonstrated that the excellent photocatalytic H2 production performance arises from the synergetic effect between the confined ultrafine CoOx nanodots and N, S heteroatom co-doped graphitic carbon. This work shows considerable improvements over MOF derived materials as non-noble cocatalysts and provides significant motivation for new photocatalyst development for improving the conversion efficiency of solar energy to chemical fuels.


 Please wait while we load your content...
Please wait while we load your content...