Direct solar thermochemical conversion of methanol into syngas via nanocatalysts at lower temperatures†
Abstract
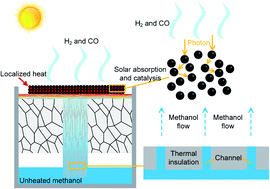
Solar thermochemical conversion is an effective way to store unstable solar energy as chemical energy in fuels; and thus, it is of great significance in the clean energy economy. Using thermochemical catalysts as the solar energy absorber directly is an advanced way to achieve high efficiency because of the cancellation of the heat resistance resulting from the heat transfer between the solar absorber and the catalyst. In this work, the photo-thermal conversion mechanism of catalyst nanoparticles in solar thermochemical processes was investigated theoretically as the first step. The multi-step temperature variations of catalyst nanoparticles with time were demonstrated. The results show that the direct solar thermochemical conversion performance is significantly enhanced by using nano-sized catalysts. Then, a CuO/ZnO/Al2O3 nanocatalyst was synthesized and utilized in a customized low-cost experimental system for the direct solar methanol decomposition. Compared with conventional systems, ours requires a lower solar concentration ratio and a lower system temperature and can be easily controlled to adjust the methanol supply rate to realize optimal solar energy utilization efficiency.


 Please wait while we load your content...
Please wait while we load your content...