Solvent selection and Pt decoration towards enhanced photocatalytic CO2 reduction over CsPbBr3 perovskite single crystals†
Abstract
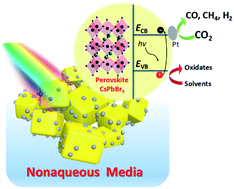
The photocatalytic reduction of CO2 into chemical fuels is one of the most important artificial photosynthesis processes, which can simulate the natural photosynthesis in green plants, and it will be a crucial procedure in the carbon neutral economy. In the current study, we report that inorganic CsPbBr3 perovskite submicron single crystals can exhibit photocatalytic reduction of CO2 directly on their surface with high stability and selectivity in several nonaqueous solvents. The solvent effects have been well studied, which finally leads to an approximately 25-fold enhancement in the photocatalytic reaction rate by substituting toluene with ethyl acetate. The loading of a Pt co-catalyst via a photochemical deposition process further doubled the electron yield rate, which finally reached 5.6 μmol g−1 h−1, thereby implying that the optimization of the reaction media and surface catalytic activity are of key importance for the superior photocatalytic performance of halide perovskite materials.


 Please wait while we load your content...
Please wait while we load your content...