Ligand regulation to prepare an Fe, N, S tri-codoped hollow carbon electrocatalyst for enhanced ORR performance and Zn–air batteries†
Abstract
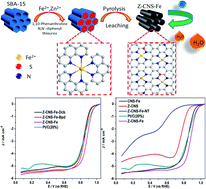
It is significant to explore high-efficiency, low-cost, and stable electrocatalysts for the oxygen reduction reaction (ORR) to substitute the precious metal platinum in electrochemical energy conversion devices. Materials with an Fe–Nx center are considered as promising catalysts to achieve this. Herein, a feasible SiO2–ZnCl2 template strategy is used to obtain nitrogen–sulfur–Fe co-doped periodic porous hollow carbon by calcining the Fe-based complexes with different ligands (1,10-phenanthroline, 2,2-bipyridine, and 1,2-dicyanobenzene). Owing to the introduction of the SBA-15 template and pore-forming agent ZnCl2, the as-prepared materials possess a large specific surface area (SSA) and a well-defined hollow structure. Most importantly, different ligands have a crucial effect on the nitrogen content of the obtained materials, which in turn directly affects the metal active sites of ORR. The appropriate selection of 1,10-phenanthroline as the ligand creates abundant Fe–Nx active sites after the pyrolysis process. Therefore, the optimal hollow carbon (denoted as Z-CNS-Fe) derived from the complex of Fe2+ and 1,10-phenanthroline exhibits better ORR performance with a half-wave potential of 0.880 V, which is 30 mV higher than that of commercial Pt/C (0.850 V). Beyond this, Z-CNS-Fe shows long-term stability and superior tolerance to methanol crossover. In addition, when Z-CNS-Fe was applied as the cathode of a primary Zn–air battery, the obtained metal–air cell provided a power density as high as 141 mW cm−2, which exceeded that of commercial Pt/C (114 mW cm−2). The high open-circuit voltage and satisfactory durability also indicate its potential practical applicability.


 Please wait while we load your content...
Please wait while we load your content...