Permselective ion electrosorption of subnanometer pores at high molar strength enables capacitive deionization of saline water†
Abstract
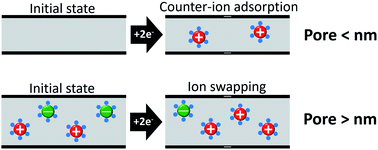
Capacitive deionization with porous carbon electrodes is an energy-efficient water treatment technique limited to the remediation of only brackish water due to the severe efficiency drop at high molar strength. Combining experiment and simulation, our work demonstrates the ability of subnanometer pores for permselective ion electrosorption, which enables capacitive deionization for saline media with high concentrations. Molecular dynamics simulations reveal the origin of permselective ion electrosorption in subnanometer pores at high molar strength. Within the subnanometer range, carbon pores with smaller size become more ionophobic and then express a higher ability of permselective ion electrosorption. This can be understood by the effects of the pore size on the microstructure of in-pore water and ions and the nanoconfinement effects on the ion hydration. These findings provide a new avenue for capacitive deionization of saline water (seawater-like ionic strength) to enable the application of highly concentrated saline media by direct use of porous carbons.


 Please wait while we load your content...
Please wait while we load your content...