Nitrogen and phosphorus modification to enhance the catalytic activity of biomass-derived carbon toward the oxygen reduction reaction†
Abstract
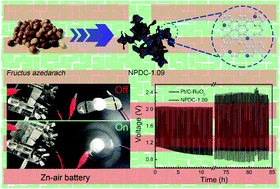
As the crucial cathode reaction of fuel cells and metal–air batteries, the oxygen reduction reaction (ORR) has always required a catalyst to improve the electrochemical reaction rate. In this article, taking Fructus azedarach as the raw material, a nitrogen–phosphorus double-doped carbon nanosheet material was synthesized through microwave-assisted hydrothermal treatment and high temperature calcination. The synthesized amorphous NPDC possessed multi-level pores and abundant edge defects. With an N : P ratio of 1.09, doped N and P achieved the best electronic synergistic effect and thus caused surrounding carbon to show high catalytic activity towards the ORR with a half-wave potential of 0.84 V, onset potential of 0.94 V and limiting current density value of 6.01 mA cm−2 in alkaline media. The NPDC-1.09 also exhibited superior stability (drop of 11% at 0.8 V after 22 h) and methanol tolerance (3.2% decline) compared to 20% Pt/C. When used in zinc–air batteries, its promising application in the field of energy storage and conversion is demonstrated by its 84 h long-term cycling stability (with a current density of 10 mA cm−2). The discovery of such an N and P dual-doped carbon structure with a substantial synergistic effect is of great significance for promoting the replacement of platinum-based catalysts with carbon-based materials.
- This article is part of the themed collection: Sustainable Energy & Fuels Cover Art


 Please wait while we load your content...
Please wait while we load your content...