Power-to-gas systems utilizing methanation reaction in solid oxide electrolysis cell cathodes: a model-based study†
Abstract
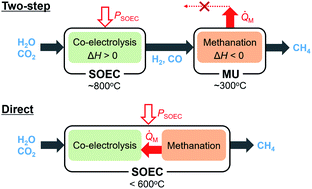
Power-to-gas is an emerging technology to convert electrical energy into chemical energy, and usually consists of two steps. First, hydrogen or syngas is produced in electrolysis cells, and then the hydrogen is used to methanate COx in catalytic reactors. Solid oxide electrolysis cells (SOECs) have the potential to integrate these two steps because methane can be directly synthesized in the SOEC cathodes. However, the advantages and drawbacks of such a direct process have not been discussed quantitatively. In the present work, we established a model using Aspen Plus to simulate the direct process. By analyzing the energy conversion processes in detail, it was clarified that the suppression of SOEC overpotentials at low temperatures is crucial. If the SOEC overpotentials are decreased at 400 °C, the energy conversion efficiency of the direct process will become higher than that of the conventional two-step process especially at small methane production rates. The superior performance can be ascribed to the recuperative heat utilization between the exothermic methanation reaction and the endothermic electrolysis reactions.
- This article is part of the themed collection: Sustainable Energy & Fuels Cover Art


 Please wait while we load your content...
Please wait while we load your content...