A cyclic electrochemical strategy to produce acetylene from CO2, CH4, or alternative carbon sources†
Abstract
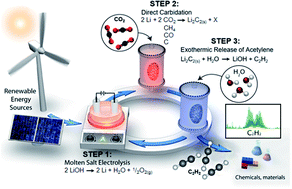
Electrochemical transformation of potent greenhouse gases such as CO2 and CH4 to produce useful carbon-based products is a highly desirable sustainability goal. However, selectivity challenges remain in aqueous electrochemical processes as selective CO2 reduction to desired products is difficult and electrochemical CH4 oxidation often proceeds at very low rates. The formation of C–C coupled products in these fields is particularly desirable as this provides a path for the production of high-value fuels and chemicals. We have developed a cyclic electrochemical strategy which can produce acetylene, a C–C coupled product, from such carbon sources and water, with favorable current density and selectivity. This strategy is exemplified with a lithium-mediated cycle: an active Li0 surface is electrochemically generated from LiOH, the newly formed Li0 reacts with a carbon source to form Li2C2, and Li2C2 is hydrolyzed to form acetylene and regenerate LiOH. We demonstrate this process primarily using CO2 gas, achieving a current efficiency of 15% to acetylene (which represents 82% of the maximum based on stoichiometric production of oxygenated byproducts, e.g. LiCO3 and/or Li2O), as verified by gas chromatography and Fourier transform infrared radiation studies. We also explore CH4, CO, and C as alternative precursors in the acetylene synthesis. Notably, the use of graphitic carbon at higher temperatures resulted in over 55% current efficiency to acetylene, with opportunity for further optimization. Importantly, this cycling method avoids the formation of common side products observed during aqueous electrochemical CO2 and CH4 redox reactions, such as H2, CO, HCO2−, or CO2. Theoretical considerations elucidate the feasibility and general applicability of this cycle and the process steps have been characterized with specific electrochemical and materials chemistry techniques. The continued development of this strategy may lead to a viable route for the sustainable production of C–C coupled carbon fuels and chemicals.


 Please wait while we load your content...
Please wait while we load your content...