Fe and W doped Bi2MoO6 nanoflakes: a promising material for efficient solar water splitting†
Abstract
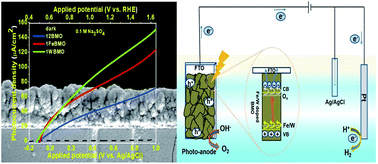
Recently, Bi2MoO6 (BMO) has been extensively investigated due to its noticeable photoelectrochemical water oxidation properties. However, the poor chemical stability and high photo-corrosion have suppressed its potential application and efficiency of solar energy conversion. In this work, BMO films have been successfully grown via a solvothermal method with different durations and the optimized synthesis conditions have been further retained to prepare Fe and W doped BMO photoanodes. Photocurrent densities as high as ≈1.81, 0.89, and 0.43 mA cm−2 at 1.23 V vs. RHE are obtained for the optimum 1% W doped, 1% Fe doped and pristine BMO, respectively, under light illumination (AM 1.5, 100 mW cm−2) in 0.1 M Na2SO4 electrolyte in the presence of 0.05 M Na2SO3 hole scavenger. A high photocurrent density of about 107.84 μA cm−2 at 1.23 V vs. RHE is obtained for the optimized W doped photoanode in the absence of 0.05 M Na2SO3 electrolyte, which is 2.18 and 1.22 times enhanced compared to those of the optimum undoped BMO and Fe doped BMO thin film photoanodes, respectively. The dopant contributed to the oxygen vacancy and film conductivity and helped both in terms of stability and PEC activity. W and Fe doped BMO exhibit considerable stability under water and sulfite oxidation conditions compared to undoped BMO in the absence of any oxygen evolution co-catalysts.


 Please wait while we load your content...
Please wait while we load your content...