X-ray absorption spectroscopy of Ba- and Cs-promoted Ru/mesoporous carbon catalysts for long-term ammonia synthesis under intermittent operation conditions†
Abstract
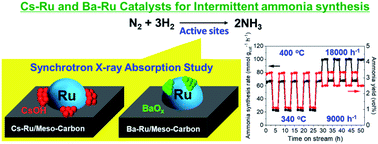
The structural characteristics of Ba- and Cs-promoted Ru catalysts supported on a mesoporous carbon material were examined by X-ray absorption spectroscopy and several characterization techniques, and correlated to their activities in long-term ammonia synthesis under intermittent operation conditions. 0.5Ba–10 wt% Ru/MPC contains barium species adhered to the surfaces of the Ru nanoparticles, creating more catalytically active sites for the dissociation of nitrogen molecules. By contrast, 2.5Cs–10 wt% Ru/MPC contains cesium hydroxides homogeneously covered on the MPC and Ru interfaces, enhancing the electronic properties of the Ru nanoparticles and consequently facilitating ammonia synthesis. As a result, the catalytic study demonstrated that ammonia synthesis rates over these promoted Ru catalysts could be finely and stably tuned within 22.5–100 mmol gcat−1 h−1 by quickly varying the reaction temperature in the range of 340–400 °C and gas hourly space velocity in the range of 9000–18 000 h−1, and the intermittent operation conditions could achieve the desired hydrogen production rates derived from a sustainable process.


 Please wait while we load your content...
Please wait while we load your content...