Synergistic effect of a noble metal free Ni(OH)2 co-catalyst and a ternary ZnIn2S4/g-C3N4 heterojunction for enhanced visible light photocatalytic hydrogen evolution†
Abstract
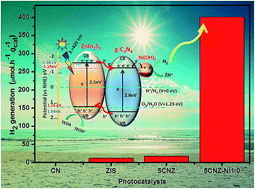
In the present study, we report the synthesis of a ternary chalcogenide (ZnIn2S4) based g-C3N4 heterojunction composite with non-noble metal Ni(OH)2 as a co-catalyst for enhanced visible light photocatalytic hydrogen generation. The optimized deposition of Ni(OH)2 (1 wt%) on ZnIn2S4/g-C3N4 resulted in 22.5 fold higher activity than that of the one without Ni(OH)2 under visible light irradiation (λ > 420 nm). And the performance for the hydrogen generation activities of the catalysts is in the order of g-C3N4 < ZnIn2S4 < ZnIn2S4/g-C3N4 ≪ ZnIn2S4/g-C3N4@Ni(OH)2. Moreover, the optimized heterojunction catalyst showed higher photocatalytic H2 generation than Pt deposition under similar experimental conditions. Therefore, the synergistic effect of the co-catalyst and ZnIn2S4/g-C3N4 helped in efficient separation of charge carriers for the selective reduction reaction and minimized charge carrier recombination, which is proven by photoluminescence studies, photocurrent measurements. Thus, the selected heterojunction composite system can be a promising photocatalyst for cleaner hydrogen energy production.


 Please wait while we load your content...
Please wait while we load your content...