Hybrid photoanodes for water oxidation combining a molecular photosensitizer with a metal oxide oxygen-evolving catalyst
Abstract
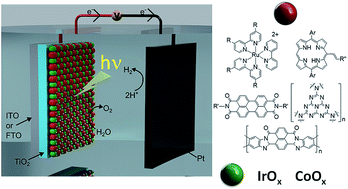
Visible light-driven water splitting performed within a photoelectrochemical cell (PEC) is one of the most investigated processes to produce hydrogen in a sustainable manner. Developing a photoanode for the water oxidation reaction (oxygen evolving reaction, OER) is considered more challenging than designing a photocathode for the water reduction reaction (hydrogen evolving reaction, HER) because the OER requires a higher overpotential and displays a higher activation barrier. Since the work of Fujishima and Honda in 1972, intense research has been devoted to the development of photoelectrodes solely based on inorganic semiconductor materials. Nevertheless, the large majority of photoanodes using inorganic semiconductors weakly absorb in the visible spectrum and present poor long-term stability. An alternative design, more recently explored, is to decouple the light absorption and the catalytic function by adsorbing a molecular photosensitizer on an n-type semiconducting electrode (generally TiO2), to promote visible light absorption, and an oxygen-evolving catalyst to promote the OER. Herein, we provide a comprehensive review of such hybrid photoanodes that associate with a molecular photosensitizer and an oxygen-evolving catalyst based on metal oxide nanoparticles. For these hybrid photoanodes, tris(bipyridine) ruthenium complexes and organic dyes such as perylenes, free-base porphyrins, polyheptazine and π-conjugated naphthalene benzimidazole polymers have been employed as molecular photosensitizers in combination with IrOx or CoOx OER catalysts. The preparation of these photoanodes, the evaluation of their photocatalytic performance for the OER, and the key factors that govern their efficiency and stability are highlighted.
- This article is part of the themed collection: Sustainable Energy and Fuels Recent Review Articles


 Please wait while we load your content...
Please wait while we load your content...