Facile room-temperature growth of nanostructured CuBi2O4 for selective electrochemical reforming and photoelectrochemical hydrogen evolution reactions†
Abstract
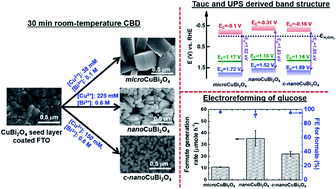
Facile, room-temperature, and surfactant-free seed-mediated chemical bath deposition was developed to directly grow CuBi2O4 nanostructures on an electrode substrate for applications in electrochemical reforming of glucose and photoelectrochemical (PEC) hydrogen generation. The metal precursor concentration and Cu2+/Bi3+ molar ratio were found to be decisive factors in determining the structure of CuBi2O4, and single crystal CuBi2O4 submicron-square columns (microCuBi2O4), nano-square columns (nanoCuBi2O4), and c-axis oriented nano-square columns (c-nanoCuBi2O4) were selected for detailed electrochemical and PEC characterization. The results of Tauc plots and ultraviolet photoelectron spectroscopy (UPS) resolved the band structure of these CuBi2O4 compounds. Mott–Schottky and UPS analyses showed that these CuBi2O4 compounds had flat-band potentials of >1.10 V vs. RHE, which makes them promising photocathodes for PEC hydrogen generation. The synthesized CuBi2O4 compounds exhibited different levels of electrocatalytic activity towards electroreforming of glucose, in the order of nanoCuBi2O4 > c-nanoCuBi2O4 > microCuBi2O4, which is attributed to the structure-dependent kinetics of the chemical reaction between glucose and electrochemically activated Cu species in CuBi2O4 at elevated anodic potentials. Additionally, all synthesized CuBi2O4 samples showed high selectivity (faradaic efficiency > 93%) for formate generation from electro-oxidative conversion of glucose. This study opens up a new avenue for the synthesis of nanostructured CuBi2O4 with promising bi-functionality towards PEC biomass reforming.


 Please wait while we load your content...
Please wait while we load your content...