Novel aqueous amine looping approach for the direct capture, conversion and storage of CO2 to produce magnesium carbonate†
Abstract
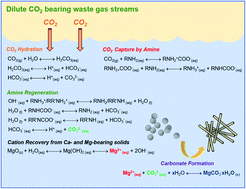
Designing novel integrated chemical pathways for the capture, conversion, and storage of CO2 is a crucial need for advancing sustainable energy conversion. The accelerated conversion of CO2 to water-insoluble and stable magnesium carbonate is a thermodynamically downhill route for permanently storing CO2. However, a practical constraint of carbon mineralization is the slow kinetics at low CO2 concentrations. In this study, we investigate a direct integrated mineralization approach, whereby CO2, amine solvents (such as monoethanolamine, MEA), water and alkaline Mg-bearing solids are reacted in a slurry reaction environment. About 70% conversion of magnesium oxide to magnesium carbonate was achieved at 50 °C after 3 hours. The looping of amine-bearing solvents between the CO2 loaded and release states, facilitates the accelerated conversion of magnesium-bearing oxides to magnesium carbonate. Hydrated magnesium carbonate and magnesium hydroxide phases were noted when less than complete conversion of magnesium oxide was achieved. The morphological features were determined using in operando ultra-small and small angle X-ray scattering (USAXS/SAXS) and grazing incidence-small angle X-ray scattering (GI-SAXS) measurements. The proposed pathway is an adaptive, low temperature, single-step approach for the direct capture, conversion and storage of CO2. By demonstrating the effectiveness of aqueous alkaline amine looping approach for the accelerated carbon mineralization of MgO, we aim to expand this approach to include heterogeneous alkaline industrial residues such as coal fly ash and steel slag and naturally occurring minerals such as magnesium silicate and calcium silicate.

 Please wait while we load your content...
Please wait while we load your content...