Organoborohydride-catalyzed Chichibabin-type C4-position alkylation of pyridines with alkenes assisted by organoboranes†
Abstract
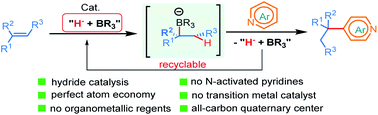
The first NaBEt3H-catalyzed intermolecular Chichibabin-type alkylation of pyridine and its derivatives with alkenes as the latent nucleophiles is presented with the assistance of BEt3, and a series of branched C4-alkylation pyridines, even highly congested all-carbon quaternary center-containing triarylmethanes can be obtained in a regiospecific manner. Therefore, the conventional reliance on high cost and low availability transition metal catalysts, prior formation of N-activated pyridines, organometallic reagents, and extra oxidation operation for the construction of a C–C bond at the C4-position of the pyridines in previous methods are not required. The corresponding mechanism and the key roles of the organoborane were elaborated by the combination of H/D scrambling experiments, 11B NMR studies, intermediate trapping experiments and computational studies. This straightforward and mechanistically distinct organocatalytic technology not only opens a new door for the classical but still far less well-developed Chichibabin-type reaction, but also sets up a new platform for the development of novel C–C bond-forming methods.


 Please wait while we load your content...
Please wait while we load your content...