Regioselective addition/annulation of ferrocenyl thioamides with 1,3-diynes via a sulfur-transfer rearrangement to construct extended π-conjugated ferrocenes with luminescent properties†
Abstract
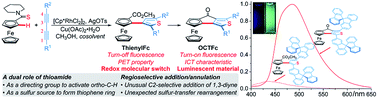
Herein a regioselective addition/annulation strategy of ferrocenyl (Fc) thioamides with alkynes to construct thienylferrocene (ThienylFc) structures, involving a rhodium-catalyzed C–H activation, an unusual C2-selective addition of 1,3-diyne, and an unexpected intramolecular sulfur-transfer rearrangement process is described. In this protocol, thioamide not only serves as a directing group to activate the ortho-C–H bond of the ferrocene, but also as a sulfur source to form the thiophene ring. The resulting carboxylic ester group after sulfur transfer can act as a linkage to construct extended π-conjugated ferrocenes (OCTFc) with luminescent properties. ThienylFc displays effective fluorescence quenching due to the photoinduced electron transfer (PET) from the Fc unit to the excited luminophore, which turns out to be a promising type of redox molecular switch. OCTFc exhibit relatively strong emission owing to their intramolecular charge transfer (ICT) characteristics. The ring-fused strategy is herein employed for the first time to construct luminescent materials based on ferrocenes, which provides inspiration for the development of novel organic optoelectronic materials, such as electroluminescent materials based on ferrocenes.


 Please wait while we load your content...
Please wait while we load your content...