Combining free energy calculations with tailored enzyme activity assays to elucidate substrate binding of a phospho-lysine phosphatase†
Abstract
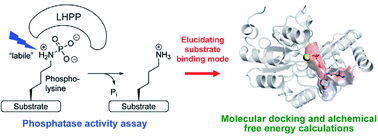
Studying enzymes that are involved in the regulation of dynamic post-translational modifications (PTMs) is of key importance in proteomics research. Such investigations can be particularly challenging when the modification itself is intrinsically labile. In this article, we elucidate the enzymatic activity of Phospholysine Phosphohistidine Inorganic Pyrophosphate Phosphatase (LHPP) towards different O- and N-phosphorylated peptides by a combined experimental and computational approach. LHPP has been previously described to hydrolyze the phosphoramidate bonds in different small molecule substrates, including phosphorylated lysine (pLys). Taking the instability of the phosphoramidate bond into account, we conducted a carefully adjusted enzymatic assay with various pLys pentapeptides to confirm enzymatic phosphatase activity with LHPP. Molecular docking was employed to explore possible binding poses of the substrates in complex with the enzyme. Molecular dynamics based free energy calculations, which are unique in their accuracy and solid theoretical basis, were further applied to predict relative binding affinity of different substrates. Comparison of simulations with experiments clearly suggested a distinct binding motif of pLys peptides as well as a very narrow promiscuity of LHPP. We believe this integrated approach can be widely adopted to study the structure and interaction of poorly characterized enzyme–substrate complexes, in particular with synthetically challenging or labile substrates.
- This article is part of the themed collection: Celebrating 10 years of Chemical Science


 Please wait while we load your content...
Please wait while we load your content...