Peptide late-stage C(sp3)–H arylation by native asparagine assistance without exogenous directing groups†
Abstract
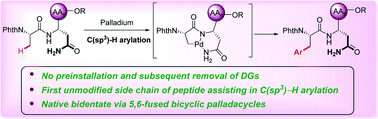
There is a strong demand for novel native peptide motifs for post-synthetic modifications of peptides without pre-installation and subsequent removal of directing groups. Herein, we report an efficient method for peptide late-stage C(sp3)–H arylations assisted by the unmodified side chain of asparagine (Asn) without any exogenous directing group. Thereby, site-selective arylations of C(sp3)–H bonds at the N-terminus of di-, tri-, and tetrapeptides have been achieved. Likewise, we have constructed a key building block for accessing agouti-related protein (AGRP) active loop analogues in a concise manner.
- This article is part of the themed collection: Editor’s Choice – Ning Jiao


 Please wait while we load your content...
Please wait while we load your content...