Site-selective aqueous C–H acylation of tyrosine-containing oligopeptides with aldehydes†
Abstract
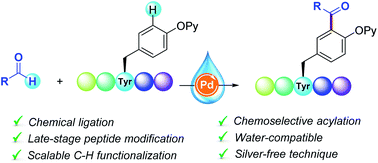
The development of useful synthetic tools to label amino acids within a peptide framework for the ultimate modification of proteins in a late-stage fashion is a challenging task of utmost importance within chemical biology. Herein, we report the first Pd-catalyzed C–H acylation of a collection of Tyr-containing peptides with aldehydes. This water-compatible tagging technique is distinguished by its site-specificity, scalability and full tolerance of sensitive functional groups. Remarkably, it provides straightforward access to a high number of oligopeptides with altered side-chain topology including mimetics of endomorphin-2 and neuromedin N, thus illustrating its promising perspectives toward the diversification of structurally complex peptides and chemical ligation.


 Please wait while we load your content...
Please wait while we load your content...