Cancer cell-selective modulation of mitochondrial respiration and metabolism by potent organogold(iii) dithiocarbamates†
Abstract
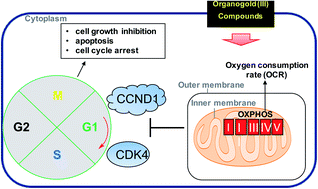
Metabolic reprogramming is a key cancer hallmark that has led to the therapeutic targeting of glycolysis. However, agents that target dysfunctional mitochondrial respiration for targeted therapy remains underexplored. We report the synthesis and characterization of ten (10) novel, highly potent organometallic gold(III) complexes supported by dithiocarbamate ligands as selective inhibitors of mitochondrial respiration. The structure of dithiocarbamates employed dictates the biological stability and cellular cytotoxicity. Most of the compounds exhibit 50% inhibitory concentration (IC50) in the low-micromolar (0.50–2.9 μM) range when tested in a panel of aggressive cancer types with significant selectivity for cancer cells over normal cells. Consequently, there is great interest in the mechanism of action of gold chemotherapeutics, particularly, considering that DNA is not the major target of most gold complexes. We investigate the mechanism of action of representative complexes, 1a and 2a in the recalcitrant triple negative breast cancer (TNBC) cell line, MDA-MB-231. Whole-cell transcriptomics sequencing revealed genes related to three major pathways, namely: cell cycle, organelle fission, and oxidative phosphorylation. 2a irreversibly and rapidly inhibits maximal respiration in TNBC with no effect on normal epithelial cells, implicating mitochondrial OXPHOS as a potential target. Furthermore, the modulation of cyclin dependent kinases and G1 cell cycle arrest induced by these compounds is promising for the treatment of cancer. This work contributes to the need for mitochondrial respiration modulators in biomedical research and outlines a systematic approach to study the mechanism of action of metal-based agents.


 Please wait while we load your content...
Please wait while we load your content...