Deoxygenative α-alkylation and α-arylation of 1,2-dicarbonyls†
Abstract
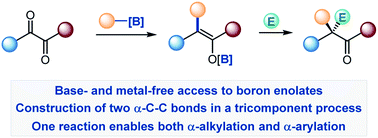
Construction of C–C bonds at the α-carbon is a challenging but synthetically indispensable approach to α-branched carbonyl motifs that are widely represented among drugs, natural products, and synthetic intermediates. Here, we describe a simple approach to generation of boron enolates in the absence of strong bases that allows for introduction of both α-alkyl and α-aryl groups in a reaction of readily accessible 1,2-dicarbonyls and organoboranes. Obviation of unselective, strongly basic and nucleophilic reagents permits carrying out the reaction in the presence of electrophiles that intercept the intermediate boron enolates, resulting in two new α-C–C bonds in a tricomponent process.
- This article is part of the themed collection: 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...