Exohedral functionalization vs. core expansion of siliconoids with Group 9 metals: catalytic activity in alkene isomerization†
Abstract
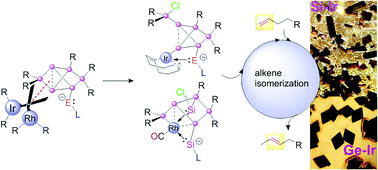
Taking advantage of pendant tetrylene side-arms, stable unsaturated Si6 silicon clusters (siliconoids) with the benzpolarene motif (the energetic counterpart of benzene in silicon chemistry) are successfully employed as ligands towards Group 9 metals. The pronounced σ-donating properties of the tetrylene moieties allow for sequential oxidative addition and reductive elimination events without complete dissociation of the ligand at any stage. In this manner, either covalently linked or core-expanded metallasiliconoids are obtained. [Rh(CO)2Cl]2 inserts into an endohedral Si–Si bond of the silylene-functionalized hexasilabenzpolarene leading to an unprecedented coordination sphere of the Rh centre with five silicon atoms in the initial product, which is subsequentially converted to a simpler derivative under reconstruction of the Si6 benzpolarene motif. In the case of [Ir(cod)Cl]2 (cod = 1,5-cyclooctadiene) a similar Si–Si insertion leads to the contraction of the Si6 cluster core with concomitant transfer of a chlorine atom to a silicon vertex generating an exohedral chlorosilyl group. Metallasiliconoids are employed in the isomerization of terminal alkenes to 2-alkenes as a catalytic benchmark reaction, which proceeds with competitive selectivities and reaction rates in the case of iridium complexes.
- This article is part of the themed collections: Spotlighting main group elements in polynuclear complexes and 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...