Comprehending the quadruple bonding conundrum in C2 from excited state potential energy curves†
Abstract
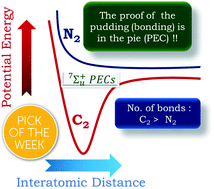
The question of quadruple bonding in C2 has emerged as a hot button issue, with opinions sharply divided between the practitioners of Valence Bond (VB) and Molecular Orbital (MO) theory. Here, we have systematically studied the Potential Energy Curves (PECs) of low lying high spin sigma states of C2, N2, Be2 and HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CH using several MO based techniques such as CASSCF, RASSCF and MRCI. The analyses of the PECs for the 2S+1Σg/u (with 2S + 1 = 1, 3, 5, 7, 9) states of C2 and comparisons with those of relevant dimers and the respective wavefunctions were conducted. We contend that unlike in the case of N2 and HC
CH using several MO based techniques such as CASSCF, RASSCF and MRCI. The analyses of the PECs for the 2S+1Σg/u (with 2S + 1 = 1, 3, 5, 7, 9) states of C2 and comparisons with those of relevant dimers and the respective wavefunctions were conducted. We contend that unlike in the case of N2 and HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CH, the presence of a deep minimum in the 7Σ+ state of C2 and CN+ suggests a latent quadruple bonding nature in these two dimers. Our investigations reveal that the number of bonds in the ground state can be determined for 2nd row dimers by figuring out at what value of spin symmetry a purely dissociative PEC is obtained. For N2 and HC
CH, the presence of a deep minimum in the 7Σ+ state of C2 and CN+ suggests a latent quadruple bonding nature in these two dimers. Our investigations reveal that the number of bonds in the ground state can be determined for 2nd row dimers by figuring out at what value of spin symmetry a purely dissociative PEC is obtained. For N2 and HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CH the purely dissociative PEC appears for the septet spin symmetry as compared to that for the nonet in C2. This is indicative of a higher number of bonds between the two 2nd row atoms in C2 as compared to those of N2 and HC
CH the purely dissociative PEC appears for the septet spin symmetry as compared to that for the nonet in C2. This is indicative of a higher number of bonds between the two 2nd row atoms in C2 as compared to those of N2 and HC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CH. Hence, we have struck a reconciliatory note between the MO and VB approaches. The evidence provided by us can be experimentally verified, thus providing the window so that the narrative can move beyond theoretical conjectures.
CH. Hence, we have struck a reconciliatory note between the MO and VB approaches. The evidence provided by us can be experimentally verified, thus providing the window so that the narrative can move beyond theoretical conjectures.
- This article is part of the themed collections: 2020 ChemSci Pick of the Week Collection and 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...