Prediction of NHC-catalyzed chemoselective functionalizations of carbonyl compounds: a general mechanistic map†
Abstract
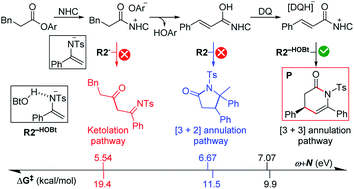
Generally, N-heterocyclic carbene (NHC) complexed with carbonyl compounds would transform into several important active intermediates, i.e., enolates, Breslow intermediates, or acylazolium intermediates, which act as either a nucleophile (Nu) or an electrophile (E) to react with the other E/Nu partner. Hence, the key to predicting the origin of chemoselectivity is to compute the activity (i.e., electrophilic index ω for E and nucleophilic index N for Nu) and stability of the intermediates and products, which are suggested in a general mechanistic map of these reactions. To support this point, we selected and studied different cases of the NHC-catalyzed reactions of carbonyl compounds in the presence of a base and/or an oxidant, in which multiple possible pathways involving acylazolium, enolate, Breslow, and α,β-unsaturated acylazolium intermediates were proposed and a novel index ω + N of the E and Nu partners was employed to exactly predict the energy barrier of the chemoselective step in theory. This work provides a guide for determining the general principle behind organocatalytic reactions with various chemoselectivities, and suggests a general application of the reaction index in predicting the chemoselectivity of the nucleophilic and electrophilic reactions.


 Please wait while we load your content...
Please wait while we load your content...