Transferring axial molecular chirality through a sequence of on-surface reactions†
Abstract
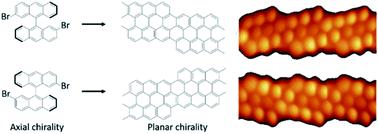
Fine management of chiral processes on solid surfaces has progressed over the years, yet still faces the need for the controlled and selective production of advanced chiral materials. Here, we report on the use of enantiomerically enriched molecular building blocks to demonstrate the transmission of their intrinsic chirality along a sequence of on-surface reactions. Triggered by thermal annealing, the on-surface reactions induced in this experiment involve firstly the coupling of the chiral reactants into chiral polymers and subsequently their transformation into planar prochiral graphene nanoribbons. Our study reveals that the axial chirality of the reactant is not only transferred to the polymers, but also to the planar chirality of the graphene nanoribbon end products. Such chirality transfer consequently allows, starting from adequate enantioenriched reactants, for the controlled production of chiral and prochiral organic nanoarchitectures with pre-defined handedness.
- This article is part of the themed collections: Editor’s Choice – Ning Jiao and 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...