Metal–metal cooperative bond activation by heterobimetallic alkyl, aryl, and acetylide PtII/CuI complexes†
Abstract
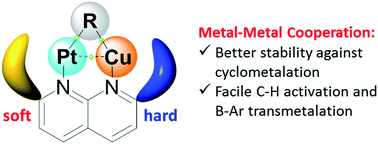
We report the selective formation of heterobimetallic PtII/CuI complexes that demonstrate how facile bond activation processes can be achieved by altering the reactivity of common organoplatinum compounds through their interaction with another metal center. The interaction of the Cu center with the Pt center and with a Pt-bound alkyl group increases the stability of PtMe2 towards undesired rollover cyclometalation. The presence of the CuI center also enables facile transmetalation from an electron-deficient tetraarylborate [B(ArF)4]− anion and mild C–H bond cleavage of a terminal alkyne, which was not observed in the absence of an electrophilic Cu center. The DFT study indicates that the Cu center acts as a binding site for the alkyne substrate, while activating its terminal C–H bond.
- This article is part of the themed collection: Celebrating five years of ChemRxiv


 Please wait while we load your content...
Please wait while we load your content...