D–π–D molecular semiconductors for perovskite solar cells: the superior role of helical versus planar π-linkers†
Abstract
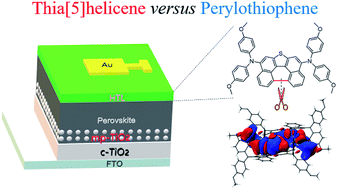
Controlling the mode of molecular packing and the size of molecular aggregates is of fundamental importance for high-performance charge transport materials in next-generation optoelectronic devices. To clarify the peculiar role of helicene as a kernel block in the exploration of unconventional organic semiconductors, in this work thia[5]helicene (T5H) is doubly aminated with electron-donating dimethoxydiphenylamine to afford T5H–OMeDPA, which is systematically compared with its perylothiophene (PET) congener (PET–OMeDPA). On the basis of the quantum theory of atoms in molecules and energy decomposition analysis of single crystals, it is surprisingly found that while π–π stacking of planar PET is stronger than that of helical T5H, this desirable effect for the charge transport of organic semiconductors is completely lost for donor–π–donor (D–π–D) type PET–OMeDPA but is retained for T5H–OMeDPA to a large extent. Consequently, the T5H–OMeDPA single-crystal presents about 5 times higher theoretical hole-mobility than PET–OMeDPA. More critically, the solution-processed racemic glassy film of T5H–OMeDPA displays a 3 times higher hole-mobility in comparison with the PET–OMeDPA counterpart, due to a larger domain of molecular aggregates. With respect to PET–OMeDPA, there is a weaker electronic coupling of helical T5H–OMeDPA with perovskites, leading to reduced interfacial charge recombination. Due to reduced transport resistance and enhanced recombination resistance, perovskite solar cells with T5H–OMeDPA exhibit a power conversion efficiency of 21.1%, higher than 19.8% with PET–OMeDPA and 20.6% with the spiro-OMeTAD control.
- This article is part of the themed collection: 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...