Electrochemical biomass valorization on gold-metal oxide nanoscale heterojunctions enables investigation of both catalyst and reaction dynamics with operando surface-enhanced Raman spectroscopy†
Abstract
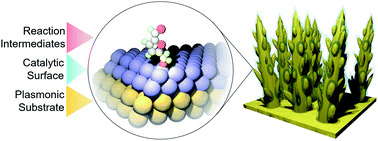
The electrochemical oxidation of biomass platforms such as 5-hydroxymethylfurfural (HMF) to value-added chemicals is an emerging clean energy technology. However, mechanistic knowledge of this reaction in an electrochemical context is still lacking and operando studies are even more rare. In this work, we utilize core–shell gold-metal oxide nanostructures which enable operando surface-enhanced Raman spectroelectrochemical studies to simultaneously visualize catalyst material transformation and surface reaction intermediates under an applied voltage. As a case study, we show how the transformation of NiOOH from ∼1–2 nm amorphous Ni layers facilitates the onset of HMF oxidation to 2,5-furandicarboxylic acid (FDCA), which is attained with 99% faradaic efficiency in 1 M KOH. In contrast to the case in 1 M KOH, NiOOH formation is suppressed, and consequently HMF oxidation is sluggish in 10 mM KOH, even at highly oxidizing potentials. Operando Raman experiments elucidate how surface adsorption and interaction dictates product selectivity and how the surface intermediates evolve with applied potential. We further extend our methodology to investigate NiFe, Co, Fe, and CoFe catalysts and demonstrate that high water oxidation activity is not necessarily correlated with excellent HMF oxidation performance and highlight catalytic factors important for this reaction such as reactant–surface interactions and the catalysts' physical and electronic structure. The insights extracted are expected to pave the way for a deepened understanding of a wide array of electrochemical systems such as for organic transformations and CO2 fixation.
- This article is part of the themed collection: Celebrating five years of ChemRxiv


 Please wait while we load your content...
Please wait while we load your content...