Aqueous zinc ion batteries: focus on zinc metal anodes
Abstract
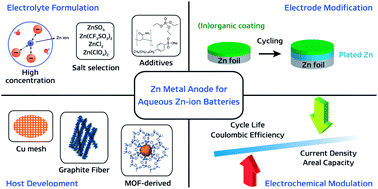
Despite the prevalence of lithium ion batteries in modern technology, the search for alternative electrochemical systems to complement the global battery portfolio is an ongoing effort. The search has resulted in numerous candidates, among which mildly acidic aqueous zinc ion batteries have recently garnered significant academic interest, mostly due to their inherent safety. As the anode is often fixed as zinc metal in these systems, most studies address the absence of a suitable cathode for reaction with zinc ions. This has led to aggressive research into viable intercalation cathodes, some of which have shown impressive results. However, many investigations often overlook the implications of the zinc metal anode, when in fact the anode is key to determining the energy density of the entire cell. In this regard, we aim to shed light on the importance of the zinc metal anode. This perspective offers a brief discussion of zinc electrochemistry in mildly acidic aqueous environments, along with an overview of recent efforts to improve the performance of zinc metal to extract key lessons for future research initiatives. Furthermore, we discuss the energy density ramifications of the zinc anode with respect to its weight and reversibility through simple calculations for numerous influential reports in the field. Finally, we offer some perspectives on the importance of optimizing zinc anodes as well as a future direction for developing high-performance aqueous zinc ion batteries.
- This article is part of the themed collections: Celebrating 15 years of exceptional research in Chemical Science, Battery science and technology – powered by chemistry, Most popular 2019-2020 review articles, Most popular 2019-2020 materials and energy chemistry articles and Celebrating Chemical Science in Korea


 Please wait while we load your content...
Please wait while we load your content...