A programmable chemical switch based on triggerable Michael acceptors†
Abstract
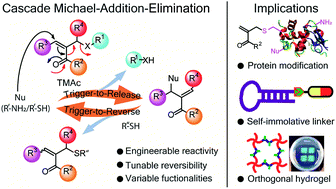
Developing an engineerable chemical reaction that is triggerable for simultaneous chemical bond formation and cleavage by external cues offers tunability and orthogonality which is highly desired in many biological and materials applications. Here, we present a chemical switch that concurrently captures these features in response to chemically and biologically abundant and important cues, viz., thiols and amines. This thiol/amine-triggerable chemical switch is based on a Triggerable Michael Acceptor (TMAc) which bears good leaving groups at its β-position. The acceptor undergoes a “trigger-to-release” process where thiol/amine addition triggers cascaded release of leaving groups and generates a less activated acceptor. The newly generated TMAc can be further reversed to liberate the original thiol/amine by a second nucleophile trigger through a “trigger-to-reverse” process. Within the small molecular volume of the switch, we have shown five locations that can be engineered to achieve tunable “trigger-to-release” kinetics and tailored reversibility. The potential of the engineerable bonding/debonding capability of the chemical switch is demonstrated by applications in cysteine-selective and reversible protein modification, universal self-immolative linkers, and orthogonally addressable hydrogels.


 Please wait while we load your content...
Please wait while we load your content...