Significantly improved electrocatalytic oxygen reduction by an asymmetrical Pacman dinuclear cobalt(ii) porphyrin–porphyrin dyad†
Abstract
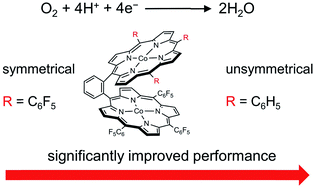
Pacman dinuclear CoII triphenylporphyrin-tri(pentafluorophenyl)porphyrin 1 and dinuclear CoII bis-tri(pentafluorophenyl)porphyrin 2, anchored at the two meso-positions of a benzene linker, are synthesized and examined as electrocatalysts for the oxygen reduction reaction (ORR). Both dinuclear Co bisporphyrins are more efficient and selective than corresponding mononuclear CoII tetra(pentafluorophenyl)porphyrin 3 and CoII tetraphenylporphyrin 4 for the four-electron electrocatalytic reduction of O2 to water. Significantly, although the ORR selectivities of the two dinuclear Co bisporphyrins are similar to each other, 1 outperforms 2, in terms of larger catalytic ORR currents and lower overpotentials. Electrochemical studies showed different redox behaviors of the two Co sites of 1: the CoIII/CoII reduction of the Co-TPP (TPP = triphenylporphyrin) site is well-behind that of the Co-TPFP (TPFP = tri(pentafluorophenyl)porphyrin) site by 440 mV. This difference indicated their different roles in the ORR: CoII-TPFP is likely the O2 binding and reduction site, while CoIII-TPP, which is generated by the oxidation of CoII-TPP on electrodes, may function as a Lewis acid to assist the O2 binding and activation. The positively charged CoIII-TPP will have through-space charge interactions with the negatively charged O2-adduct unit, which will reduce the activation energy barrier for the ORR. This effect of Co-TPP closely resembles that of the CuB site of metalloenzyme cytochrome c oxidase (CcO), which catalyzes the biological reduction of O2. This work represents a rare example of asymmetrical dinuclear metal catalysts, which can catalyze the 4e reduction of O2 with high selectivity and significantly improved activity.
- This article is part of the themed collection: 2019 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...